- Blog
- About
- Contact
- Sketchup pro 2022 full
- Angel stone rpg with friends
- Skim and scan
- 2016 quicken error codes
- Open shared contacts outlook 2016
- Mods for x3 albion prelude
- 24 tv serial free download
- A wizards lizard soul thief 0-22-0
- Unistall ms remote desktop app on mac os
- Unbound book
- How do i get groove agent 4 to use my groove agent se kits
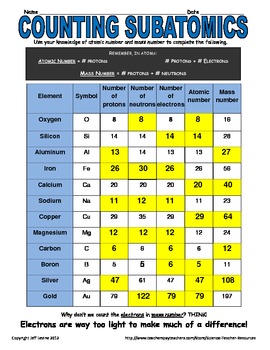
- Subatomic particles list
- Microsoft office 2019 for mac free download
- Ubar east point ga
- What is the best mp4 to mp3 converter free download

The classical theory also states that there is another particle, the photon, which has no mass and is responsible for electromagnetic waves, as in light. In classical atomic theory, atoms are composed of three subatomic particles: electrons, protons, and neutrons, the first two of which have an electrical charge, while neutrons are neutral. The isotopes of protium, deuterium, and tritium (from left to right).

Hydrogen, H: with an atomic number of 1, this is the most abundant element in the universe.See the following examples of atoms belonging to different elements, including their symbols and atomic numbers: Different elements have different numbers of protons. Camacho, StudySmarter.Ītoms are classified by their atomic number, which identifies the number of protons found in the nucleus. An atom, with protons and neutrons displayed in green and the orbiting electrons in red. Electrons move around the nucleus.įigure 1. Protons and neutrons are the particles that exist in the atom's nucleus. Atoms of different chemical elements have different numbers of neutrons, protons, and electrons, which gives them their distinctive properties. Methane, which is used as fuel, consists of four hydrogen atoms and one carbon atom.Ītoms, the basic blocks of chemistry, are the smaller units of matter that can have a neutral charge.Oxygen, which we breathe every day, consists of two oxygen atoms.Water, which consists of two atoms of hydrogen and one atom of oxygen.Total Internal Reflection in Optical Fibre.Newton’s and Huygens’ Theories of Light.Einstein's Theory of Special Relativity.Electromagnetic Radiation and Quantum Phenomena.Magnetic Flux and Magnetic Flux Linkage.
